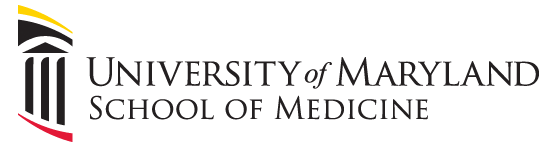Category: Critical Care
Keywords: critically ill, ED, boarding, outcome (PubMed Search)
Posted: 11/25/2025 by Quincy Tran, MD, PhD
Click here to contact Quincy Tran, MD, PhD
Settings: this is a meta-analysis of 17 observational studies about boarding of critically ill patients in US Emergency Departments. All studies were from urban, academic centers.
Participants:
Outcome measurement: all cause mortality, as reported by the authors of the original studies.
Study Results:
Discussion:
Conclusion:
Critically ill patients boarding in the U.S. Emergency Departments were associated with a non-statistically signi?cant increase in odds of mortality and hospital length of stay compared to non-boarded patients
Htet NN, Walker JA, Jafari D, Rech MA, Hintze T, Moran M, Bai J, Dinh K, Essaihi A, Wilairat S, Huddleson B, Tran QK. Outcomes of boarding critically ill patients in U.S. EDs: A systematic review and meta-analysis. Am J Emerg Med. 2025 Oct 17;99:339-347. doi: 10.1016/j.ajem.2025.10.036. Epub ahead of print. PMID: 41151219.
Category: Critical Care
Keywords: bicarbonate, metabolic acidosis, renal replacement therapy, acute kidney injury (PubMed Search)
Posted: 11/25/2025 by Jessica Downing, MD
Click here to contact Jessica Downing, MD
The role of sodium bicarbonate in the treatment of severe acidemia has been controversial, with some studies suggesting no benefit, and others indicating that it may help reduce need for renal replacement therapy (RRT) and even improve mortality. The BICARICU-2 Trial was an open-label multicenter RCT conducted in France that evaluated the impact of a bicarb infusion among patients with metabolic acidosis and moderate to severe AKI.
There was no difference in 90 day mortality, but patients in the bicarb group were less likely to be started on RRT (38% vs 47% in the control group) using pre-defined criteria for RRT initiation, and had a 50% lower rate of bloodstream infections. Patients in the bicarb group who were started on RRT met criteria for RRT later than those in the control group (median 31h vs 15.5h).
Study Details:
Patient Population:
Intervention:
RRT Triggers:
Jung B, Jabaudon M, De Jong A, Bitker L, Audard J, Klouche K, Sarton B, Guitton C, Lasocki S, Rieu B, Canet E, Jeantrelle C, Roquilly A, Mayaux J, Verdonk F, Pottecher J, Ferrandiere M, Riu B, Garcon P, Assefi M, Detouche P, Forel JM, Roger C, Bourenne J, Jacquier S, Bougon D, Rolle A, Corne P, Benchabane N, Richard JC, Asehnoune K, Chanques G, Reignier J, Belafia F, Fosset M, Huguet H, Futier E, Molinari N, Jaber S; BICARICU-2 Study Group. Sodium Bicarbonate for Severe Metabolic Acidemia and Acute Kidney Injury: The BICARICU-2 Randomized Clinical Trial. JAMA. 2025 Oct 29:e2520231. doi: 10.1001/jama.2025.20231. Epub ahead of print. PMID: 41159812; PMCID: PMC12573113.
Category: Critical Care
Posted: 11/18/2025 by Caleb Chan, MD
Click here to contact Caleb Chan, MD
This is an actual patient case:
65 y/o pt intubated for hemoptysis and started on nebulized transexamic acid. Overnight, the pt is found to have severe breath stacking/auto-PEEPing and consequently is started on neuromuscular blockade. The pt has no history of asthma or COPD and the ETT is clear without obstruction.
Ventilator waveforms are as shown. What is the issue?
Explanation:
On expiration, the ventilator pressure (and the pressure curve waveform on the ventilator) should drop to the set PEEP (10 cm H2O in this case) immediately. This is true regardless of whether it is volume control, pressure control, PRVC etc. For this patient, the pressure curve is not dropping to the set PEEP immediately on expiration, rather, it slowly decays and does not even reach the set PEEP before the beginning of the next breath. This is not due to a patient issue, but rather an obstruction at the level of the ventilator. In particular, an obstruction in the expiratory limb of the tubing where flow returns to the ventilator from the patient. TXA is known to crystallize on the expiratory filter which can cause this type of obstruction if it is not changed frequently enough, preventing the pressure from dropping to PEEP and the patient from fully exhaling.
In this case, the obstruction was localized to the expiratory filter based on the ventilator waveforms and the filter was exchanged. The waveforms normalized, the patient had no obstruction or breath stacking, the neuromuscular blockade discontinued, and the patient was subsequently extubated without issue.
Category: Critical Care
Keywords: Critical Care, Surgical Critical Care, Fellowship, Training, Medical education, Emergency Medicine-Critical Care, EM-CC (PubMed Search)
Posted: 11/12/2025 by William Teeter, MD
Click here to contact William Teeter, MD
This study surveyed 111 emergency medicine (EM) trainees to identify factors influencing their choice of critical care (CC) fellowship pathways, particularly surgical critical care (SCC). Respondents included 42 fellows and 69 residents, with most pursuing anesthesiology or medicine CC; only 15 intended SCC.
Key determinants of pathway selection were:
Limited exposure to EM-SCC during residency was noted—only 28% had access to such fellowships, and 42% interacted with surgical intensivists, despite 41% envisioning SCC practice.
Intellectual appeal ranked highest for entering CC, above job prospects or lifestyle.
Fellowship components most valued were:
While descriptive, the authors noted many respondents cited the "preliminary surgical year" as a reason that the Surgical Critical Care pathway is less attractive.
The authors conclude that respondents pursued a career in CC for "intellectual appeal and desire for additional expertise" and that improving EM-SCC matriculation requires targeted interventions.
Hynes AM, Carver TW, Owodunni OP, et al. Attracting Emergency Medicine-Trained Residents to Surgical Critical Care: The Implications From a Nationwide Survey of Emergency Medicine Trainees Interested in Critical Care. Crit Care Med. 2025 Oct 31. doi: 10.1097/CCM.0000000000006935.
Category: Critical Care
Keywords: Sepsis, Shock, Hypotension, Fluids, Ultrasound, Vasopressors (PubMed Search)
Posted: 11/4/2025 by Mark Sutherland, MD
Click here to contact Mark Sutherland, MD
Another month, another study of hemodynamic targets in sepsis… The age-old questions: is a MAP > 65 a good target for everybody, or should we individualize? Should we just give a bolus of fluids to everyone and then move to pressors, or should this strategy change patient to patient?
Huet et al have a preprint that'll appear in Intensive Care Medicine looking at this question in 517 patients. I can't reprint it here due to copyright (follow link below, go to full PDF and scroll to figures at bottom if curious), but basically their algorithm was 1) check if patient is fluid responsive via either echo or swan, 2) give fluid if yes, 3) do something else (pressors) if no.
Importantly the differences were not statistically significant, but they found a strong, nearly significant, trend towards benefit on SOFA score, ICU and hospital LOS in the “personalized therapy” group (also of note, these are dubious as patient oriented outcomes). The sickest patients (by SOFA) showed the most benefit.
Bottom Line: The “personalized hemodynamic therapy” literature continues to show a modest benefit of using tools like echo (e.g. LVOT VTI) to determine if the patient is fluid responsive (or fluid tolerant) and NOT give fluid (instead using pressors) if that is not the case, but for now there's relatively limited support for hyper-personalized approaches like varying MAP goals or otherwise mixing up your strategy. Some day we'll likely find a more nuanced approach, but for now I think a reasonable strategy in critically ill septic patients is to use ultrasound to determine if the patient needs fluid, if yes give fluid and reassess, and if not move to pressors, to maintain a MAP > 65.
Category: Critical Care
Keywords: Cardiac arrest, norepinephrine, re-arrest, advantage, epinephrine (PubMed Search)
Posted: 11/1/2025 by Robert Flint, MD
Click here to contact Robert Flint, MD
A scoping review of literature involving norepinephrine use during cardiac arrest associated with a shockable rhythm found:
-evidence in animal and signal in human trials of improved myocardial and cerebral blood flow
-a suggestion of less re-arrest
There is not enough evidence comparing epinephrine to norepinephrine however this would be an excellent area of research with a theoretical advantage to norepinephrine.
Bouman, S.J., Baldussu, E., Franssen, G.H. et al. The effects of norepinephrine in shockable cardiac arrest, a scoping review. Scand J Trauma Resusc Emerg Med 33, 155 (2025). https://doi.org/10.1186/s13049-025-01480-6
Category: Critical Care
Keywords: Ventilator, autoPEEP, asthma, COPD, obstructive lung disease (PubMed Search)
Posted: 10/28/2025 by Zachary Wynne, MD
Click here to contact Zachary Wynne, MD
Bottom line:
If a ventilated patient exhibits at least one of: persistent end expiratory flow, unequal inspiratory and expiratory flow-time areas, or ineffective breath triggers; autoPEEP must be evaluated by performing an end-expiratory hold.
If present, ventilator settings should be changed to maximize exhalation time.
In critically ill patients with obstructive lung disease, intubation and mechanical ventilation is often a last resort as it does not fix the underlying pathology of small airway disease. While many complications can arise, the most feared complication is autoPEEP.
What is autoPEEP?
AutoPEEP is excess air trapping in the lungs because the patient has insufficient time to fully exhale. Patients at highest risk include those with obstructive lung pathology due to their increased resistance (from bronchospasm) and sometimes increased compliance (such as in emphysema).
However, it is possible for any patient to develop autoPEEP depending on the amount of time they have to exhale. As respiratory rate increases, the expiratory time decreases proportionally if inspiratory time is kept constant. Ultimately, autoPEEP can lead to rapidly increasing intrathoracic pressures causing decreased preload leading to hemodynamic instability and potentially cardiac arrest. These elevated pressures also place the patient at significant risk of barotrauma/volutrauma.
How do I find it?
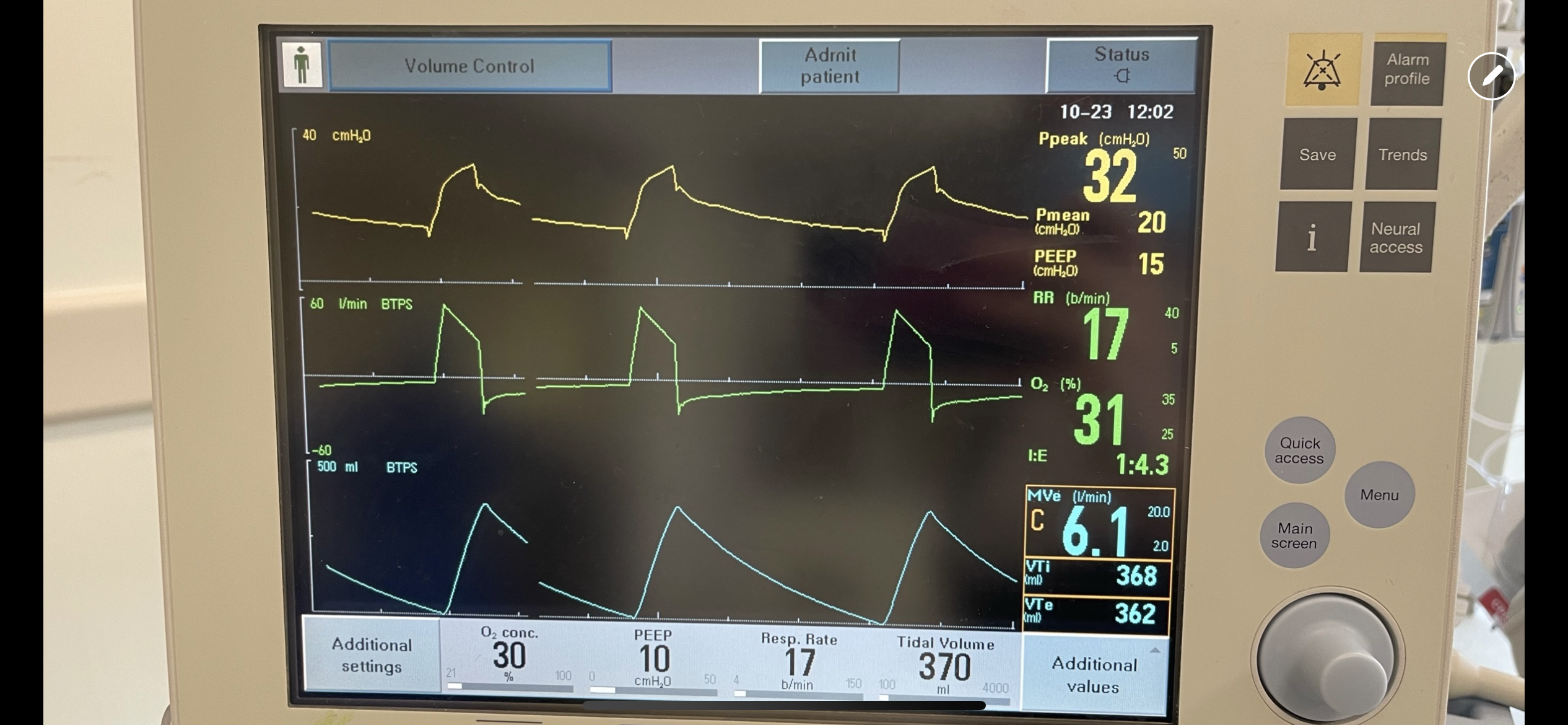
There are several signs on the ventilator waveforms for autoPEEP. Some patients may only exhibit one of the following signs of autoPEEP. They are demonstrated in the attached pictures in various ventilator modes.
Image A. Persistent end expiratory flow on the flow-time curve (middle curve) - demonstrated by the expiratory limb of the flow curve not returning to zero (remains negative)
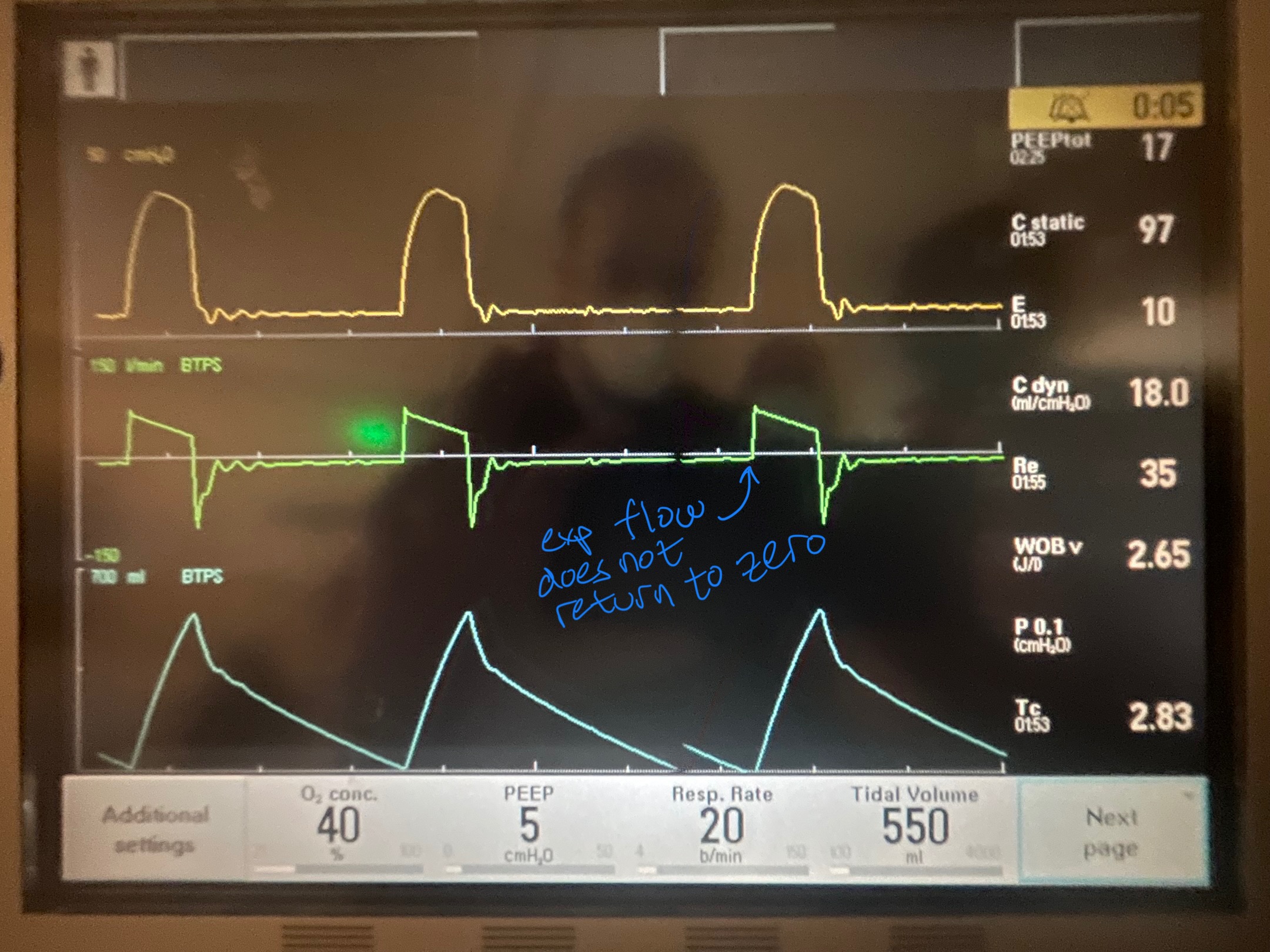
Image B. Unequal inspiratory and expiratory volumes on the flow-time curve (area of flow curve inspiratory limb does not equal area of flow curve expiratory limb)
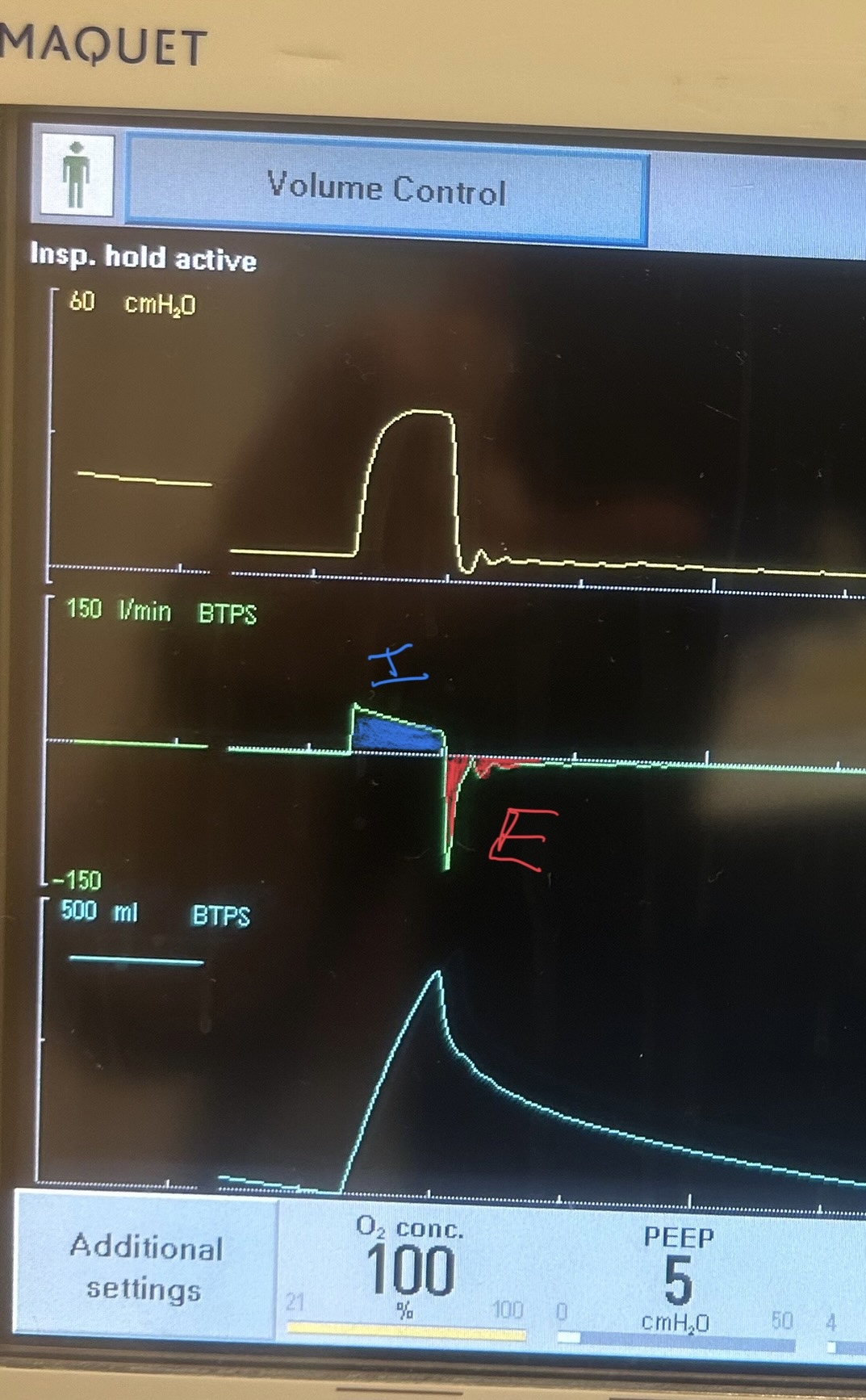
Image C. Ineffective triggering (seen on flow-time curve; patient has to perform more work to reach trigger threshold when autoPEEP is present; they are sometimes unable to trigger a breath)
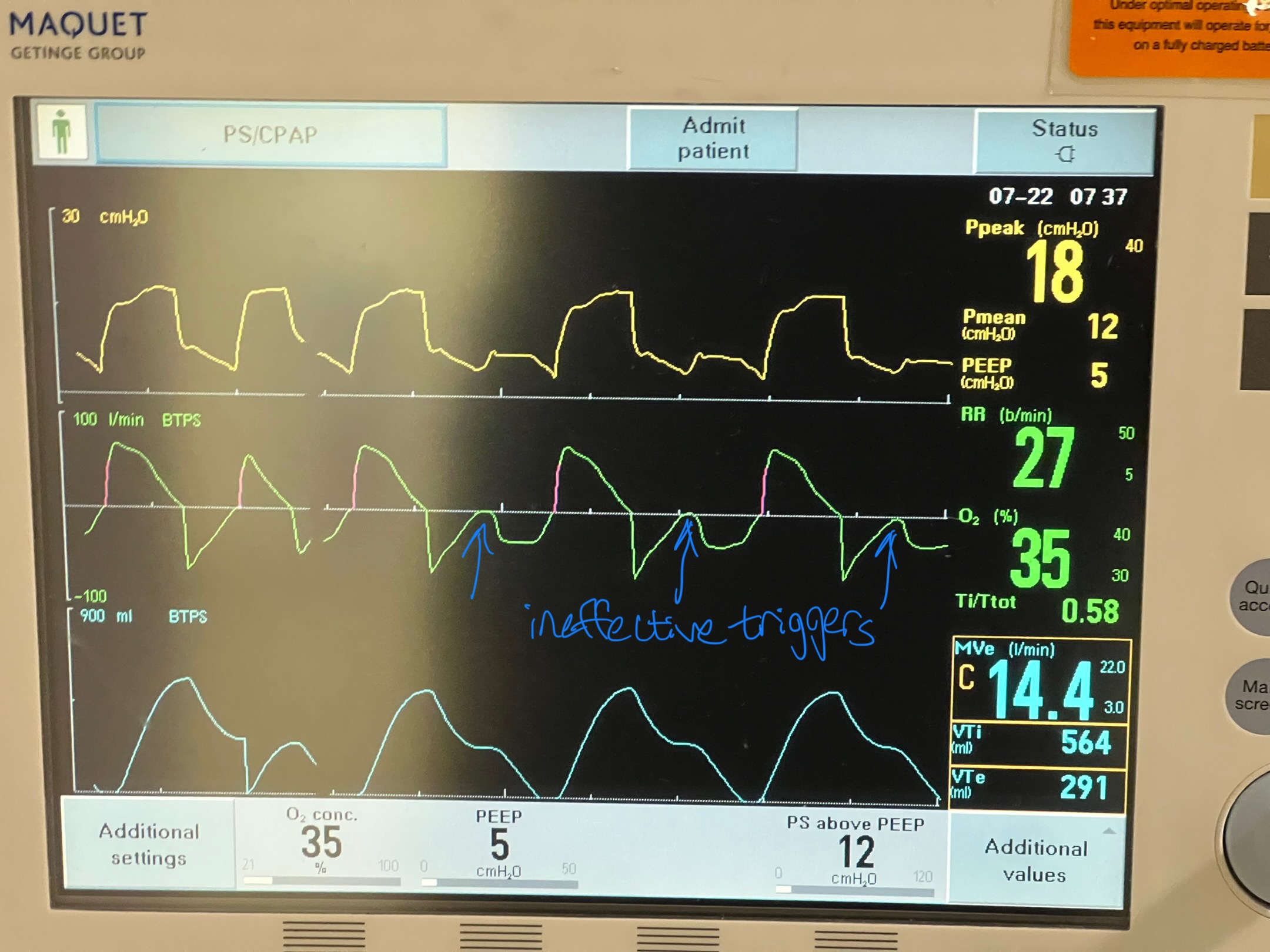
If any of these are present, an end-expiratory hold maneuver should be performed.
Image D - End-expiratory hold maneuver (done if patient is passive on the ventilator) - the pressure-time curve will begin at ventilator set PEEP and reach total PEEP at the end of the maneuver. The difference between total PEEP and set PEEP is autoPEEP.
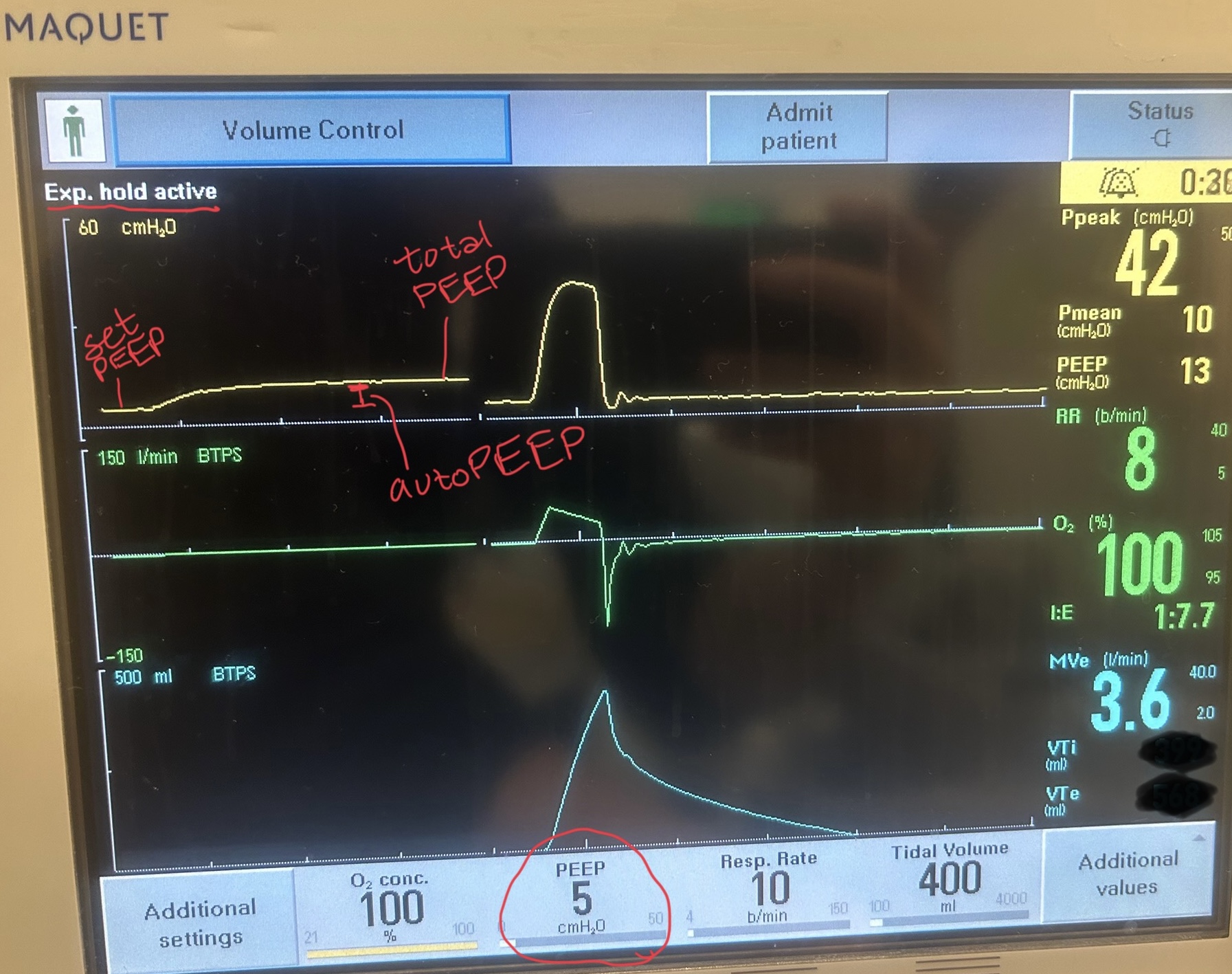
If autoPEEP is present, ventilator changes to allow for more exhalation time should be made. The most effective change is by decreasing the respiratory rate though small improvements can be made by changing the inspiratory time and tidal volume. Appropriate bronchodilator therapy, sedation, and treatment of underlying pathology is also critical in these patients.
For more information on autoPEEP, check out this post by Dr. John Greenwood discussing autoPEEP on MarylandCCProject with video demonstrations!
Category: Critical Care
Posted: 10/21/2025 by Mike Winters, MBA, MD
Click here to contact Mike Winters, MBA, MD
Check for Elevated ICP in the Post-ROSC Patient
Long B, Gottlieb M. Emergency medicine updates: Managing the patient with return of spontaneous circulation. Am J Emerg Med. 2025; 26-36.
Category: Critical Care
Keywords: delirium, ICU, acetylcholinesterase inhibitor (PubMed Search)
Posted: 10/14/2025 by Quincy Tran, MD, PhD
(Updated: 4/30/2026)
Click here to contact Quincy Tran, MD, PhD
Delirium is common among critically ill patients. Some of the common Acetylcholinesterase inhibitors (AChEI), rivastigmine, donepezil, have been used to prevent delirium in ICU patients. However, their efficacy was just recently re-examined in a meta-analysis of only Randomized Control Trials.
Ten studies and 731 patients were included- 365 in the treatment (AChEI) group and 366 in the control group.
AChEI was associated with lower occurrence of delirium (RR 0.68, 95% CI 0.47-0.98, p=0.039. However, there was no significant difference in the delirium duration (mean difference -0.16 day, 95% CI -0.95 to 0.62 day, p=0.23). There was no difference in delirium severity nor length of hospital stay.
Among the medication, interestingly, rivastigmine 4.5 mg/day significantly reduced delirium occurrence (RR = 0.61 [0.39– 0.97]) and severity (SMD = –0.33 [–0.58 to –0.08]), as well as length of hospital stay (MD = –1.29 [–1.87 to –0.72]).
Discussion:
This meta-analysis was well-conducted.
The cholinergic dysregulation—especially elevated acetylcholinesterase activity—can lead to the imbalance between attention and cognition, contributing to delirium in ICU patients. Thus, the use of AChEI and reduction of occurrence of delirium proves that acetylcholine deficiency may be associated with delirium among ICU patients.
Subgroup analysis showed that prophylactic use of AChEI was associated with significant reduction of delirium duration. Thus, further studies are needed to define which populations will benefit from AChEI.
Conclusion:
AChEIs are effective in reducing occurrence of delirium, but they did not affect delirium duration, severity or hospital LOS.
Pipek LZ, Pascual GS, Nascimento RFV, Silva GD, Castro LH. Acetylcholinesterase Inhibitors for Delirium Prevention: A Systematic Review and Meta-Analysis. Crit Care Med. 2025 Oct 1;53(10):e2054-e2061. doi: 10.1097/CCM.0000000000006786. Epub 2025 Aug 5. PMID: 40758382.
Category: Critical Care
Keywords: acute respiratory failure, hypercapnia, hypercarbia, COPD, AE-COPD, noninvasive ventilation, high flow nasal cannula (PubMed Search)
Posted: 10/7/2025 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Q: Can you use high flow nasal cannula (HFNC) to manage acute hypercapnic respiratory failure?
A: It probably depends.
Background: While we now frequently utilize HFNC as an initial therapy for most acute hypoxic respiratory failure, its appropriateness in managing acute respiratory failure with hypercarbia has historically been opposed. With more recent data indicating that HFNC may be as good as noninvasive ventilation (NIV) for management of hypercapnia as well, this seemed like a good time to point out a few things:
The RENOVATE trial was a larger multicenter randomized noninferiority trial looking at HFNC vs NIV in all-comer acute respiratory failure, summarizing that HFNC was noninferior in the primary composite outcome of death + intubation at 7 days.
BUT this conclusion is not clearly supported in the smaller COPD (or acute cardiogenic pulmonary edema) subgroup:
What does seem to be clear across studies that HFNC has the capacity to clear some CO2 and is by and large better tolerated than facemask NIV.
Bottom Line: For mild-moderate acute COPD exacerbations with patient intolerance or exclusion criteria for NIV therapy, trialing HFNC is a reasonable option. For patients with severe acute or acute on chronic hypercapnia, as indicated by a [pseudo-arbitrary] pH < 7.25 and PaCO2 >70-80, noninvasive ventilation should be your go-to… or be ready to promptly intubate if/when the high flow fails.
Category: Critical Care
Keywords: sepsis, septic shock, omeprazole, proton pump inhibitor, anti-inflammatory (PubMed Search)
Posted: 9/30/2025 by Quincy Tran, MD, PhD
Click here to contact Quincy Tran, MD, PhD
Settings: multinational, randomized, double- blind, placebo-controlled clinical trial conducted in 17 centers in Italy, Russia, and Kazakhstan
Participants: A total of 307 ICU patients with sepsis or septic shock. Patients who were likely to die (APACHE II > 65 points) were excluded.
Treatment group: 80 mg bolus of omeprazole at randomization, at 12 hours and infusion of 12 mg/hour for 72 hours. Total dose of 1024 mg.
Outcome measurement: primary outcome of the study was organ dysfunction measured as the mean daily SOFA score during the first 10 days. Secondary outcomes were antibiotics-free days at 28 days; all-cause mortality at 28 days
Study Results:
Discussion:
Conclusion:
In sepsis patients, Esomeprazole did not re- duce organ dysfunction, despite demonstrating in vivo immunomodulatory effects
Monti G, Carta S, Kotani Y, Bruni A, Konkayeva M, Guarracino F, Yakovlev A, Cucciolini G, Shemetova M, Scapol S, Momesso E, Garofalo E, Brizzi G, Baldassarri R, Ajello S, Isirdi A, Meroi F, Baiardo Redaelli M, Boffa N, Votta CD, Borghi G, Montrucchio G, Rauch S, D'Amico F, Pace MC, Paternoster G, Vitale F, Giardina G, Labanca R, Lembo R, Marmiere M, Marzaroli M, Nakhnoukh C, Plumari V, Scandroglio AM, Scquizzato T, Sordoni S, Valsecchi D, Agrò FE, Finco G, Bove T, Corradi F, Likhvantsev V, Longhini F, Konkayev A, Landoni G, Bellomo R, Zangrillo A; PPI-SEPSIS Study Group. A Multinational Randomized Trial of Mega-Dose Esomeprazole as Anti-Inflammatory Agent in Sepsis. Crit Care Med. 2025 Aug 1;53(8):e1554-e1566. doi: 10.1097/CCM.0000000000006720. Epub 2025 May 29. PMID: 40439536.
Category: Critical Care
Keywords: compartment syndrome, abdomen, critically ill (PubMed Search)
Posted: 9/24/2025 by Robert Flint, MD
(Updated: 9/28/2025)
Click here to contact Robert Flint, MD
This review article reminds us that abdominal hypertension and compartment syndrome need to remain on our differential diagnosis for critically ill and injured patients. Pressure is measured with an intra-bladder catheter. Normal pressure is 5-7 mm HG. Sustained over 12 mm Hg is hypertension and sustained over 20 mm Hg is compartment syndrome.
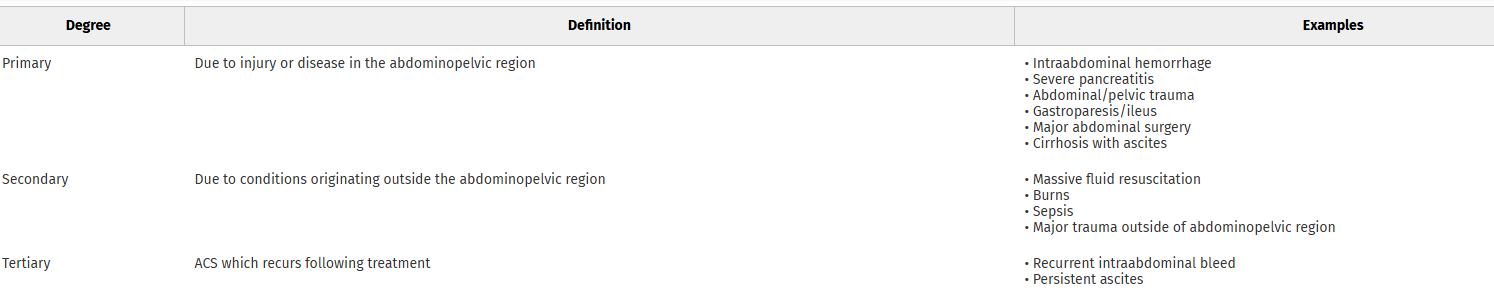
Arcieri, Talia R. MD; Meizoso, Jonathan P. MD, MSPH, FACS. Intraabdominal hypertension and abdominal compartment syndrome: What you need to know. Journal of Trauma and Acute Care Surgery 99(4):p 504-513, October 2025. | DOI: 10.1097/TA.0000000000004603
Category: Critical Care
Keywords: Oxygenation, Lateral Positioning, Hypoxia (PubMed Search)
Posted: 9/9/2025 by Mark Sutherland, MD
(Updated: 4/30/2026)
Click here to contact Mark Sutherland, MD
We've got supine positioning and prone positioning... what about something in-between? Ye et al studied 2,159 patients coming out of anesthesia in a PACU after extubation. As sedation wore off, they placed one group in lateral decubitus, and left the other group supine. The lateral decubitus group had less hypoxia, a higher lowest SpO2, and required fewer airway rescue maneuvers.
Of note, the investigators didn't compare lateral or supine to prone positioning, which is often felt to be the best position for oxygenation (depending on patient characteristics and pathophysiology). And of course, this study represents a very specific scenario quite different from the ED (PACU patients post-extubation), so it's not clear how broadly extrapolatable this is. But this does add to the argument that supine is a poor position for oxygenating patients.
Bottom Line: If your supine patient is oxygenating marginally and you want a small bump without going all the way to prone positioning, consider lateral positioning. May make the most sense for procedural sedation and post-extubation patients in terms of similarity to this particular study.
Ye H, Chu LH, Xie GH, Hua YJ, Lou Y, Wang QH, Xu ZX, Tang MY, Wang BD, Hu HY, Ying J, Yu T, Wang HY, Wang Y, Ye ZJ, Bao XF, Wang MC, Chen LY, Wang XX, Zhang XB, Huang CS, Wang J, Lu YP, Luo FQ, Zhou W, Wang CG, Cheng H, Liu WJ, Luo J, Wu YQ, Li RR, Wang D, Hou LQ, Shi L, Zhang J, Wang K, Pi X, Zhou R, Yang QQ, Wan PL, Li H, Wu SJ, Song SW, Cui P, Shu L, Islam N, Fang XM. Effect of lateral versus supine positioning on hypoxaemia in sedated adults: multicentre randomised controlled trial. BMJ. 2025 Aug 19;390:e084539. doi: 10.1136/bmj-2025-084539. PMID: 40829895; PMCID: PMC12362200.
Category: Critical Care
Keywords: Critical Care, oxygen, ventilator, SaO2 (PubMed Search)
Posted: 9/2/2025 by Zachary Wynne, MD
Click here to contact Zachary Wynne, MD
What is the ideal oxygen saturation goal for a mechanically ventilated patient? Literature over the past decade has led away from the perfect 100% oxygen saturation due to its association with worse patient outcomes across many disease states. It is theorized that excess oxygen leads to free radical production causing a lung injury pattern. However, there is no clear guidance for the ideal range of oxygen saturation goals, particularly in the mechanically ventilated patient, despite a meta-analysis and several recent trials.
UK-ROX Trial - JAMA - June 2025
Question: Does an oxygen saturation goal of 88-92% lead to a lower 90-day mortality compared to usual care?
Population: 16,500 mechanically ventilated adult patients in 97 ICU’s across the UK, excluded patients on ECMO
Intervention: Goal oxygen saturation of 88-92%, using the lowest possible FiO2
Control: Usual care, defined as oxygen supplementation at the discretion of the treating physician (no limits set to FiO2 or SaO2)
Outcomes:
Bottom Line:
Ideal oxygenation targets remain elusive. UK-ROX adds to the growing literature of oxygenation targets in mechanically ventilated patients but does not clearly show that lower oxygen saturation targets lead to improved ICU outcomes. In your emergency department ICU boarder, avoid a 100% oxygen saturation to prevent oxygen toxicity associated lung injury and consider an oxygen saturation goal of 90-96% (88-92% if history of COPD).
Martin DS, Gould DW, Shahid T, Doidge JC, Cowden A, Sadique Z, Camsooksai J, Charles WN, Davey M, Francis-Johnson A, Garrett RM, Grocott MPW, Jones J, Lampro L, Mackle DM, O'Driscoll BR, Richards-Belle A, Rostron AJ, Szakmány T, Warren A, Young PJ, Rowan KM, Harrison DA, Mouncey PR; UK-ROX Investigators. Conservative Oxygen Therapy in Mechanically Ventilated Critically Ill Adult Patients: The UK-ROX Randomized Clinical Trial. JAMA. 2025 Aug 5;334(5):398-408. doi: 10.1001/jama.2025.9663. PMID: 40501321; PMCID: PMC12163715.
Category: Critical Care
Keywords: hyperbaric, dive medicine, evaluation, (PubMed Search)
Posted: 8/31/2025 by TJ Gregory, MD
(Updated: 4/30/2026)
Click here to contact TJ Gregory, MD
You've encountered it at ABEM General Hospital, but now a SCUBA diver actually comes into your ED and you're concerned for DCS. What next?
Evaluation:
Symptom nature and timing are key in detailed history. Transient neurocognitive symptoms at depth suggest nitrogen narcosis or oxygen toxicity. Neurological symptoms within 10 minutes of surfacing suggest AGE. Widely variable symptoms within 24 hours of surfacing suggest DCS. Symptom onset greater than 24 hours suggests alternative diagnosis (still discuss with Hyperbaric Medicine or DAN).
Thorough physical exam. DCS may manifest only as localized pain. Look for marine envenomation or trauma to the area.
Neurological exam including detailed sensation and ataxia/balance - get the patient on their feet!
Unbiased differential. E.g. DCS may cause chest pain or SOB, but divers still have heart attacks. SCUBA setting may raise alert for AGE, but divers still have strokes. People go to the tropics to dive, but they also eat local fish (Scombroid and Ciguatera for a future pearl).
Management:
Early consult to Hyperbaric Medicine. In settings with no such team available, a good resource is the Divers Alert Network (DAN) Emergency Hotline at 1-919-684-9111
100% O2 via NRB or highest available delivery. You're not titrating to spO2, you're creating a diffusion gradient for tissue inert gas washout.
IV access and isotonic Fluids. PO if tolerable and unable to obtain IV access.
NSAIDs unless otherwise contraindicated. No special regimen. Standard dosing Ibuprofen or Naproxen are fine. Toradol is ok if limitations to PO.
Horizontal positioning in bed for AGE. Trendelenburg is not recommended.
Manage end organ effects as applicable. E.g. Spinal DCS may yield bladder retention requiring foley
Give consideration to activity specific considerations: hypothermia, restrictive clothing, etc
IV lidocaine has mixed evidence for neuroprotection in AGE. Discuss with Hyperbaricist before starting.
Pre-hospital considerations:
Transport should occur via ground or pressurized air transit capable of 1.0 ATA (sea level) cabin pressure. If non-pressurized aircraft transport is absolutely necessary, maintain continuous oxygen supplementation and altitude less than 2000 feet. This also applies to the inter-hospital setting.
O2 delivery by best means available to include SCUBA regulator mouthpiece or even a rebreather apparatus if present.
PO fluids if tolerable and no IV available.
https://pubmed.ncbi.nlm.nih.gov/8378877/
[Clinical aspects, pathophysiology and therapy of decompression sickness]Abstract The primary treatment of decompression illnesses (arterial gas embolism and all types of decompression sickness) is recompression therapy, combined with hyperbaric oxygen breathing. It is essential to initiate treatment as soon as the symptoms arise. However, prior to hyperbaric oxygen therapy--particularly with any delay in starting recompression--specific supportive therapy for ...pubmed.ncbi.nlm.nih.gov
Decompression Sickness - Injuries; Poisoning - MSD Manual Professional Edition
Decompression Sickness - MSD ManualsSymptoms and Signs of Decompression Sickness Severe symptoms may manifest within minutes of surfacing, but in most patients, symptoms begin gradually, sometimes with a prodrome of malaise, fatigue, anorexia, and headache. Symptoms occur within 1 hour of surfacing in approximately 50% of patients and by 6 hours in 90%. Rarely, symptoms can manifest 24 to 48 hours after surfacing, particularly ...www.msdmanuals.com
Category: Critical Care
Posted: 8/26/2025 by Mike Winters, MBA, MD
(Updated: 4/30/2026)
Click here to contact Mike Winters, MBA, MD
Predicting NIV Failure
Frat JP, et al. Noninvasive respiratory supports in the ICU. Intensive Care Med. 2025; 51:1476-89.
Category: Critical Care
Keywords: vasopressors, vasopressin, septic shock (PubMed Search)
Posted: 8/18/2025 by Jessica Downing, MD
(Updated: 8/19/2025)
Click here to contact Jessica Downing, MD
Norepinephrine (NE) is widely accepted as the first-line vasopressor for the management of septic shock, supported by the Surviving Sepsis Guidelines (1). The use of vasopressin as a second-line agent is also supported by the Surviving Sepsis Campaign, although the appropriate “triggers” for its addition remain vague. The SSG recommend adding vasopressin when NE infusion rates reach 0.25-0.6 mcg/kg/min, citing a catecholamine-sparing effect and potentially improved mortality (1, 2, 3).
What’s New?
The OVISS study (“Optimal vasopressin initiation in septic shock. The OVISS reinforcement learning study”) used machine learning to derive and internally validate a set of rules guiding the addition of vasopressin to NE for patients with septic shock using multiple databases of patient encounters across multiple institutions (4).
The machine learning model suggested initiation of vasopressin in more patients (87% vs 31%), earlier, and in less sick patients than was seen to be common practice:
Practice consistent with the above triggers was associated with decreased odds of in-hospital mortality (AOR 0.81, 95% CI 0.73-0.91).
Limitations
This was not a prospective study or RCT and was only internally validated. Using databases may limit the number of clinical variables available for analysis, and clinical judgment (how the patient looks) is not reflected.
Bottom Line
Consider adding vasopressin for patients with vasodilatory shock with low MAP despite NE >0.2mcg/kg/min and adequate fluid resuscitation, though more evidence is needed for a strong recommendation. As dual-pressor therapy may be riskier via peripheral IV and vasopressin does not have a direct antidote for extravasation, consider central line placement when adding vasopressin (5,6)
Category: Critical Care
Keywords: intubation, sedation, rapid sequence intubation, RSI, rocuronium, succinylcholine, etomidate, ketamine, propofol (PubMed Search)
Posted: 8/12/2025 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Whether you agree or disagree that “roc rocks and succ sucks,” evidence shows that approximately 3-4% of intubated patients experience awareness while paralyzed [1,2], and more of these patients are in the rocuronium subgroup [2,3,4]. Rocuronium acts in a dose-dependent fashion; the relatively standard 1-1.2 mg/kg in emergency department rapid sequence intubation (RSI) can result in a duration of paralysis can of up to 60-90 minutes. Commonly used sedatives in RSI, however, such as etomidate and ketamine, wear off quickly, before before rocuronium's paralytic effects have abated.
A recent single-center study showed that the majority of patients (60%) receiving rocuronium for paralysis during rapid sequence intubation (RSI) received no additional sedation until more than 15 minutes after induction, whether in the ED or ICU [5].
Patients experiencing awareness during paralysis with post-traumatic stress disorder [1,2] including distress from being restrained, feeling procedures, and feeling of impending death.
Bottom line: Start appropriate dose sedation promptly after RSI, especially with rocuronium, to avoid short- and long-term distress to patients.
Category: Critical Care
Keywords: diarrhea, ICU, mechanically ventilated (PubMed Search)
Posted: 8/4/2025 by Quincy Tran, MD, PhD
Click here to contact Quincy Tran, MD, PhD
Have you ever wondered what happened to your mechanically ventilated patients who developed diarrhea. Apparently, a multicenter study involving 2650 patients from 44 ICUs in the US, Canada and Saudi Arabia investigated the prevalence of diarrhea among these patients.
This study was the Editor’s choice for June 2025.
Results:
The mean age for the population was 59.8 (16.5) years, with APACHE II Score of 22.0 (7.8). Up to 61% of the patients received vasopressors or inotropes on day 1, which mean these patients are relatively ill.
Up to 60% of patients had diarrhea during their ICU stay, with 15% had diarrhea on day 1 or 2.
Initiating laxatives and antibiotics (who in the ICU would not receive vitamin V and Vitamin Z?) were associated with increased risk of diarrhea: HR for laxatives 1.28 (1.13–1.44), p<0.001; HR for antibiotics 1.41 (1.20–1.67), P< 0.001.
Furthermore, enteral feeding with high/moderate protein concentration was also associated with diarrhea (HR 1.13, 1.00-1.28, P=0.045.
Not surprisingly, diarrhea was associated with higher number of C. Diff testing.
Although patients with diarrhea were associated with longer ICU stay (15 [10-23] days) vs. those without diarrhea (8 [6-12] days), it was not associated with higher mortality (HR 0.70, 95% CI 0.57-0.86, P<0.001)
Discussion:
1. The authors did not report the rates of positive C. Diff. infection in these patients during ICU stay, although they did report that for another study in this population, the rate of positive C. Diff. infection during ICU stay was 2.2%. If only 2.2% had C. Diff. infection while up to 60% had diarrhea. Consequently, for every 30 patients with diarrhea, only one patient had C. Diff. infection. Therefore, do we have to check C. Diff. in those ICU patients with diarrhea every time?
2. The authors hypothesized that patients with diarrhea had longer ICU stay and lower mortality because they survived long enough to develop diarrhea. Thus, diarrhea is bad for clinicians, but may not be too bad for patients?
Conclusion:
Diarrhea is common among invasively ventilated patients. Patients who received laxatives, antibiotics, enteral feeding with high protein amount are at higher risk for diarrhea.
Dionne JC, Johnstone J, Heels-Ansdell D, Tahvildar Khazaneh P, Zytaruk N, Clarke F, Hand L, Millen T, Dechert W, Porteous R, Auld F, Hunt M, Campbell E, Bentall T, Campbell T, Smith O, Rose L, Arabi YM, Duan E, Wilcox ME, McIntyre L, Rochwerg B, Karachi T, Adhikari NK, Charbonney E, St-Arnaud C, Kristof A, Khwaja K, Marquis F, Zarychanski R, Golan E, Cook D; PROSPECT Research Coordinators Group, the PROSPECT Investigators and the Canadian Critical Care Trials Group. Diarrhea in Mechanically Ventilated Patients: A Nested Multicenter Substudy. Crit Care Med. 2025 Jun 1;53(6):e1247-e1256. doi: 10.1097/CCM.0000000000006667. Epub 2025 Apr 3. PMID: 40459385.
Category: Critical Care
Posted: 7/29/2025 by Caleb Chan, MD
Click here to contact Caleb Chan, MD
PEEP is often titrated up along with FiO2 to increase oxygen saturation. Although the potential negative hemodynamic effect of high PEEP is often recognized, it is important to also note that high PEEP can also paradoxically worsen oxygen saturation.
The primary physiologic explanation for this phenomenon in a patient with pulmonary disease is due to the varying impact of PEEP on the intra- vs. extra-alveolar blood vessels. PEEP preferentially distends more normal/compliant lung which causes compression of intra-alveolar vessel at excessively high levels of PEEP. This causes pulmonary blood to be diverted to areas of lower vascular resistance (e.g. consolidated lung which is less distended due to its worsened compliance) and lower VQ matching. Essentially, blood flow to normal/healthy lung is decreased and is instead increased to diseased lung, worsening hypoxemia.
Bottom line:
High PEEP can potentially worsen hypoxemia and should be considered as an etiology for worsening oxygen saturation, particularly when the hypoxemia is out of proportion to the patient’s radiographic findings.
Çoruh B, Luks AM. Positive end-expiratory pressure. When more may not be better. Annals ATS. 2014;11(8):1327-1331.